Hexacoordinated Carbon Atom Detected
Scientists from Freie Universität Berlin Demonstrated Existence of Unusual Organic Molecule with a Hexacoordinated Carbon Atom
№ 052/2017 from Mar 14, 2017
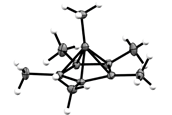
Scientists from Freie Universität Berlin have isolated an organic molecule whose structure seems to contradict fundamental principles of organic chemistry. "The doubly positively charged ion of the organic molecule hexamethylbenzene with the formula C6(CH3)62+ has a pyramidal structure with a pentagon as the base area," explains Dr. Moritz Malischewski of Freie Universität, who led the research. Benzene compounds normally have a flat six-membered ring consisting of six carbon atoms. "We are confident that this molecule will at some point be included in the textbooks of organic chemistry," says Prof. Konrad Seppelt, who was involved in the study. At the current state of research, it appears that application possibilities for the newly discovered carbon compound are unlikely. "The existence of the compound is limited to solutions in superacids at low temperatures," says Malischewski. If it comes in contact with air humidity, the compound is destroyed immediately. The structural demonstration of this molecule, however, is an important contribution to a better understanding of chemical bonding in organic molecules. The research findings were published in the prestigious journal Angewandte Chemie.
In the 1970s Dutch chemists had already correctly recognized the five-ring structure, but they were unable to isolate the compound to demonstrate a final proof of the structure, according to the scientists. Seppelt says it is particularly unusual that the carbon atom that forms the tip of the pyramid is bound to six other carbon atoms, since carbon can normally form bonds to a maximum of four adjacent atoms. Malischewski adds that this rule of the four-bond of carbon still applies. With single crystal X-ray diffraction, the scientists were able to show that the five bonds of the tip of the pyramid are significantly longer and thus weaker than normal carbon-carbon single bonds. They were also able to confirm this with quantum chemical calculations. – Malischewski explains that a single crystal is a particularly high-quality crystal in which all the molecules are arranged regularly in a uniform crystal lattice – and the molecular structure can be determined with X-ray diffraction on the single crystal.
Press Image
Molecular structure of C6(CH3)62+ in the crystal
Credit: Malischewski / Seppelt
The Publication
M. Malischewski, K. Seppelt (2017): “Crystal Structure Determination of the Pentagonal-Pyramidal Hexamethylbenzene Dication C6(CH3)62+”, in: Angew. Chem. Int. Ed. 2017, 56, pp. 368-370. http://onlinelibrary.wiley.com/doi/10.1002/anie.201608795/full, DOI: 10.1002/anie.201608795
Contact
Dr. Moritz Malischewski, Institute of Biology and Chemistry, Freie Universität Berlin, Tel.: +49 30 838-906534, Email: moritz.malischewski@fu-berlin.de